Global take on small business news
Provided by AGPHistologics' Gentle Kylon® Biopsy Brush Poised to be the New Standard of Care for Endocervical Biopsy
I have been using the Soft-ECC® sampler for over a decade at different clinical institutions. It reliably picks up tissue for a more adequate endocervical sample than traditional ECC samplers.”
ANAHEIM, CA, UNITED STATES, May 5, 2026 /EINPresswire.com/ -- Histologics LLC, based in Anaheim, California attended the Annual Meeting of the American Society of Colposcopy and Cervical Pathology in Orlando, FL from April 23-26, 2026. The meeting featured findings by the NIH showing that Histologics’ Kylon® biopsy brushes enabled researchers to discover pre-malignant lesions in the endocervical canal and that carcinoma in-situ is present in the endocervix significantly more than previously thought.— Michelle Khan MD, MPH
Histologics is dedicated to train more physicians and allied health professionals who screen, diagnose, and treat cervical cancer precursors and cancer in the “Compassionate Colposcopy®” approach. Neal Lonky MD, MPH, public health and cervical cancer prevention expert, Founder and CEO of Histologics LLC, Clinical Professor of Obstetrics and Gynecology at the University of California Irvine, Former Elected Member of the Board of Directors of the Southern California Permanente Medical Group, and multi-patented inventor of the Kylon® device technology concluded “As a longstanding practicing physician in a very busy practice at Kaiser Permanente and training residents in Ob/Gyn procedure-based care, I think it is daunting when patients ask for new technology before the clinicians who perform colposcopy can be informed and trained. There are thousands of doctors and nurses who use and prefer them now after a decade, but we have more work to do to orient and train the rest. I don’t want my clinical colleagues blindsided, which sometimes happens, before the physicians or insurers “approve” them in whatever clinical site they practice. I do think the time is now for a “standard of care” transformation.”
Please view our webinar at https://histologicswh.com/resources/webinars.html.
Michelle Khan MD, MPH, a leader in the field of Gynecology and specifically cervical cancer prevention, colposcopy guided precursor diagnosis, and therapy, and a Clinical Professor at the Stanford University School of Medicine was an early adopter of the Kylon® biopsy devices in her colposcopy practice. Her longstanding experience led her to state “I have been using the Soft-ECC® sampler for over a decade at different clinical institutions. It reliably picks up tissue for a more adequate endocervical sample than traditional ECC samplers. I have also noted better tolerance of endocervical curettage by patients when I am using this device. I am starting to use the SoftBiopsy® more often and also finding it helpful in situations where traditional forceps doesn’t grasp the tissue adequately. I also see less bleeding compared with traditional biopsy sampling.”
In a recent training webinar series available online at the Histologics Women’s Health website (https://histologicswh.com/resources/webinars.html), Praveena Yetur MD, FCAP, FASCCP, Sr. Director, Pathologist and Medical Science Liaison, Women’s Health at LabCorp said “ It is exciting to be a part of patient care continuum with patient centered new innovations that give the best samples and patient experience with least discomfort. The SoftBiopsy® and Soft-ECC® samples are abundant and diagnostic even to perform ancillary studies like p16, Ki-67, etc… as needed. Also, processing of these samples in the lab is simple. The brush tissue biopsy sample results correlate well with associated Pap, HPV results, and excisional LEEPs (loop excision specimens).”
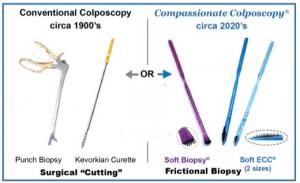
Differences between Sterile Stainless Steel Surgical Biopsy Devices and Single-Use Sterile Kylon® Brush Biopsy Devices
Traditional steel biopsy devices use surgical excision can be perceived by patients as traumatic. Opportunities to biopsy lesions are tempered by the fear of pain and bleeding and time needed to control bleeding. Reusable steel devices carry the risk of cross contamination due to the potential of incomplete cleaning and sterilization for repeated use. They can trap particles of tissue, require maintenance, and are relatively expensive to purchase and maintain. Kylon® devices are for single use and disposable carrying no-risk of cross-contamination, are gentle on the patient, efficient to use and low cost per device. The quality of the biopsy, the ability to thoroughly remove larger lesions not simply biopsy a portion, ease to perform biopsies rapidly, gentle patient experience, and the willingness of the patient to return makes this an optimal tool in the “Value-Based Care” trend emerging in the USA and the world.
About Kylon® and Histologics LLC
Histologics is a medical device manufacturer with a mission to compassionately save lives, and is dedicated to advancing gentle, effective tissue management solutions for women’s health, advanced wound care, and veterinary medicine. Its proprietary, patented, and award-winning Kylon® technology platform is a hooked brush array medical fabric that can safely and compassionately be used to precisely and gently remove tissue from the body for diagnostics and therapeutic removal of diseased tissue. Histologics provides clinicians with innovative cost-effective tools that enhance procedural efficiency, accuracy, and patient comfort. Histologics products have been adopted by leading academic, government, HMO, hospitals, private practice settings, and home settings nationwide.
A compendium of research and clinical evidence over the last 15 years can be found at: https://www.histologics.com/resources/research-summary.html
For more information, visit https://histologics.com or contact Histologics Women’s Health Division at 888-738-9757.
Neal M Lonky
Histologics LLC
+1 8882352275
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
TikTok
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.